-
PPMA condemns allegations against efficacy of Pakistani drugs
KARACHI: The Pakistan Pharmaceutical Manufacturers Association (PPMA) on Tuesday strongly condemned and categorically dismissed recent media reports in which it was alleged that the drugs produced in Pakistan often “do not meet international standards”. The above remarks were made during a session of the Senate Standing Committee on National Health Services, Regulations, and Coordination which…
-
Import of pharma products: PCDA voices concern over new infrastructure cess

KARACHI: The Pakistan Chemist and Druggist Association (PCDA) has raised serious concerns over the imposition of infrastructure cess at the import stage, imposed by the Excise, Taxation & Narcotics Control Department of Sindh. The PCDA emphasised the detrimental impact this levy has on the import of critical pharmaceutical products. According to the spokesperson of PCDA…
-
Sales Tax Rules, 2006: KTBA seeks inclusion of pharma sector under Rule 18A

KARACHI: The Karachi Tax Bar Association (KTBA) has formally requested the inclusion of the pharmaceutical sector under Rule 18A of the Sales Tax Rules, 2006. In a letter addressed to the Member IR, Policy, the KTBA highlighted recent changes to the tax regulations and their implications for the pharma industry. The request comes in light…
-
3-day Pharma Asia Exhibition concludes

KARACHI: The three-day 21st Pharma Asia Exhibition, the premier pharmaceutical exhibition in the region, successfully concluded Thursday at the Expo Centre, here on Thursday. The event, organized by E-Commerce Gateway, featured Deputy Mayor Karachi, Salman Abdullah Murad, as the chief guest, marking a significant milestone in the evolution of Pakistan’s pharmaceutical industry. Running from August…
-
BIOSECURE Act heads for possible vote in House of Representatives next week

After running up against a roadblock earlier this summer, the controversial BIOSECURE Act could gain new momentum next week. The U.S. House of Representatives is slated to consider the legislation during the week of Sept. 9, according to a website maintained by the congressional chamber. The bill was introduced in January and seeks to halt…
-
GSK posts COPD win for Nucala as Sanofi-Regeneron rival Dupixent nears delayed FDA decision
GSK is heading into the weekend with a win for its respiratory blockbuster Nucala—but for now, the details are slim. On Friday, GSK said that its IL-5 antibody Nucala plus inhaled maintenance therapy lowered the annualized rate of moderate or severe exacerbations among chronic obstructive pulmonary disease (COPD) patients for up to 2 years. In…
-
BF Biosciences enters into agreement with Lucky Core Industries

Pakistan’s pharmaceutical company BF Biosciences Limited (BFBL) has entered into a contract manufacturing and supply agreement with Lucky Core Industries Limited (LCI). The development was shared by Ferozsons Laboratories Limited, the parent company of BFBL, in a notice to the Pakistan Stock Exchange (PSX) on Wednesday. “BF Biosciences Limited (BFBL), a subsidiary of Ferozsons Laboratories…
-
Ferozsons Laboratories’ subsidiary BF Biosciences launches human insulin
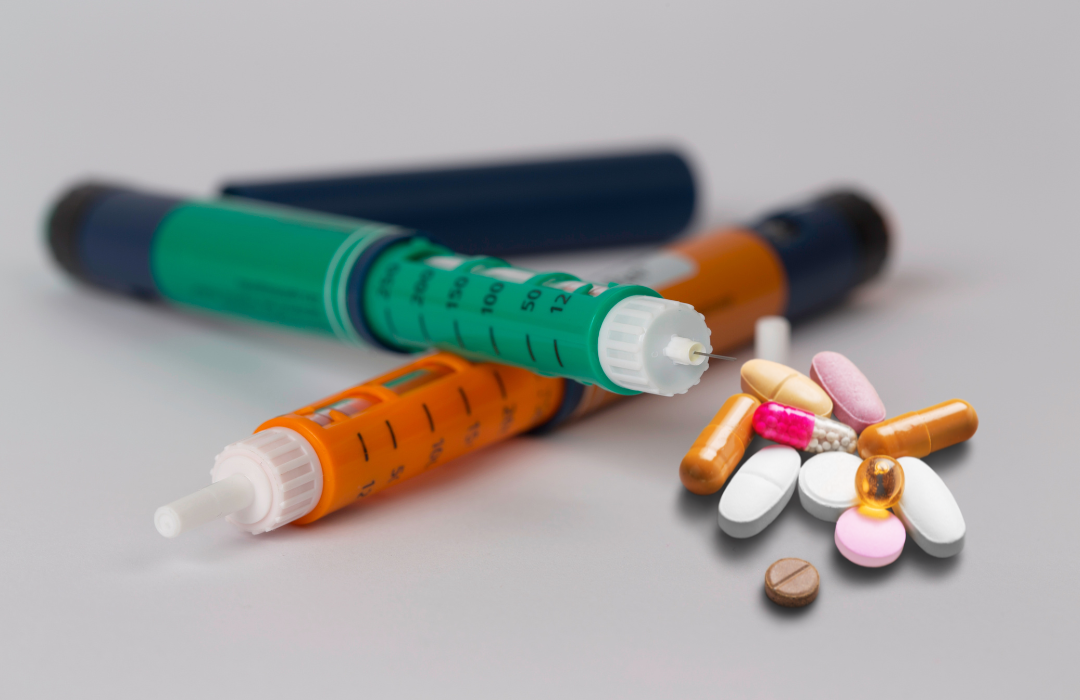
Pakistan’s pharmaceutical company BF Biosciences Limited (BFBL) has successfully launched a human insulin under the brand name ‘Ferulin’. The development was shared by Ferozsons Laboratories Limited, the parent company of BFBL, in its notice to the Pakistan Stock Exchange (PSX) on Thursday. “We are pleased to inform you that BF Biosciences Limited, a subsidiary of…
-
Pakistan’s pharma company BF Biosciences looks to raise at least Rs1.38bn through IPO

Pakistan’s pharmaceutical company BF Biosciences Limited (BFBL) is planning an initial public offering (IPO) at the Pakistan Stock Exchange (PSX) with an aim to raise at least Rs1.375 billion ($4.94 million) through the issuance of 25 million shares at a floor price of Rs55 per share. Brokerage house Arif Habib Limited has been appointed as…
-
BF Biosciences gets DRAP nod for brownfield expansion

Pakistan’s pharmaceutical company BF Biosciences Limited (BFBL) has secured approval from the Drug Regulatory Authority of Pakistan (DRAP) for brownfield expansion. The development was shared by its parent Ferozsons Laboratories Limited in a notice to the Pakistan Stock Exchange (PSX) on Monday. “We are pleased to inform that BF Biosciences Limited, a subsidiary of Ferozsons…