-
FDA Expands Apple Juice Recall Over Arsenic Concerns: Products Sold at Walmart, Walgreens, Aldi, and More Impacted

The FDA has expanded its apple juice recall due to concerns about arsenic levels. Refresco Beverages, Inc. recalled an additional 133,500 cases of its 100% apple juice after discovering arsenic levels slightly above the FDA’s limit of 10 parts per billion (ppb). The affected products were sold at major retailers like Walgreens, Aldi, Walmart, and…
-
The National Pharmaceutical Alliance Secures Clean Sweep Victory in PPMA Elections

National Pharmaceutical Alliance Dominates PPMA Elections for 2024-26 Under the guidance and leadership of prominent figures including Mian Asad Shuja-ur-Rehman (Chairman), Khawaja Shahzeb Akram, Ch. Yousaf Ali, Mian Khalid Misbah-ur-Rehman, Shafique A. Abbasi, Dr. Tahir Azam, Ehtisham-ul-Haq, Aamir Saleem Butt, Dr. Faisal Khokhar, and Dr. Riaz Ahmad from Lahore, alongside Ansar Farooq Ch., Arshad Mahmood,…
-
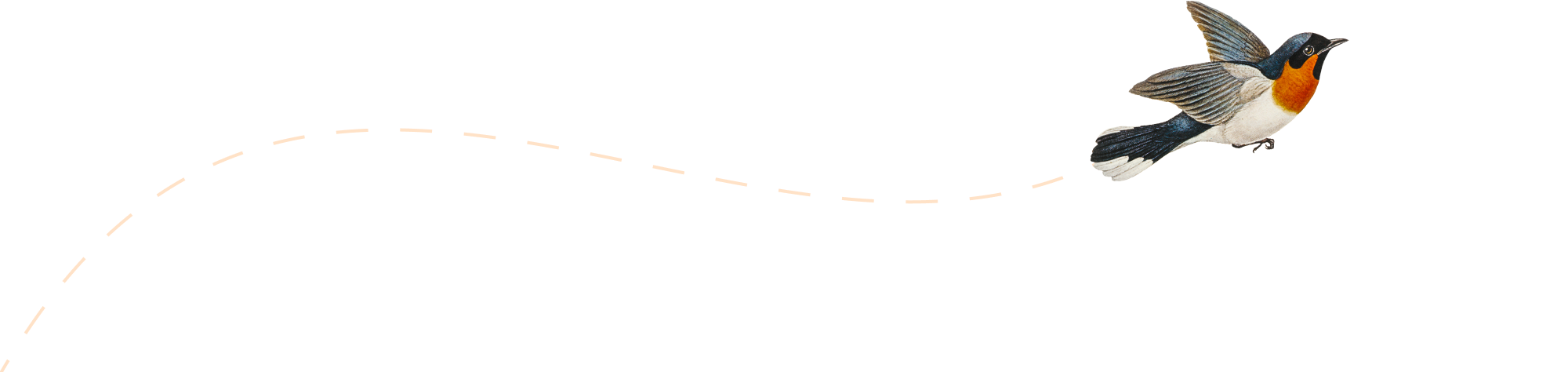
Africa CDC Calls for Urgent Mpox Response Support Amid Rising Cases and Deaths
Introduction: The Africa Centres for Disease Control and Prevention (Africa CDC) has issued an urgent appeal for increased support from Western nations in combating the ongoing mpox outbreak. With 107 deaths and 3,160 new cases reported in just one week, the agency is pushing for stronger global cooperation to meet its $600 million funding target.…
-
The Urgent Need for Reform in Pakistan’s Pharmaceutical Sector

Pakistan’s pharmaceutical industry plays a pivotal role in the country’s healthcare system, yet it is burdened by outdated laws and regulations that stifle its growth and threaten public health. The most pressing issue is the Drug Act of 1976, which, despite being over four decades old, continues to govern the pharmaceutical landscape, leading to inefficiencies,…
-
Authorities Ordered to Withdraw Seven Drug Batches Immediately
ISLAMABAD – In an effort to cleanse the drug market of counterfeit and substandard pharmaceuticals, the Drug Regulatory Authority of Pakistan (DRAP) has launched a major crackdown. Authorities have been instructed to promptly remove seven batches of drugs from distributors, pharmacies, and hospitals. According to a DRAP alert, the Directorate of Drugs Control (DDC) Punjab…
-
Government Initiates Crackdown on Counterfeit and Contaminated Drugs

ISLAMABAD: The government has launched a major crackdown on counterfeit and Contaminated drugs. The Drug Regulatory Authority of Pakistan (DRAP) has issued a directive to halt the use of products identified as spurious by drug testing laboratories. This notice, released on Friday, applies to healthcare professionals including physicians, pharmacists, nurses, and procurement managers at hospitals…
-
DRAP issues recall alert for five drug products

The Drug Regulatory Authority of Pakistan (DRAP) has announced an urgent recall of five liquid drug products found to contain harmful contaminants. These products, including Cestonil Plus Syrup, Texcol DM 10mg/5ml Syrup, Speczine 5mg/5ml Syrup, Aphylin Syrup, and Zolint Suspension, have been identified as containing ethylene glycol and di-ethylene glycol, both of which are toxic…
-
Regulator Suspends Entod Pharma’s Eye Drop Approval Over Unapproved Claims
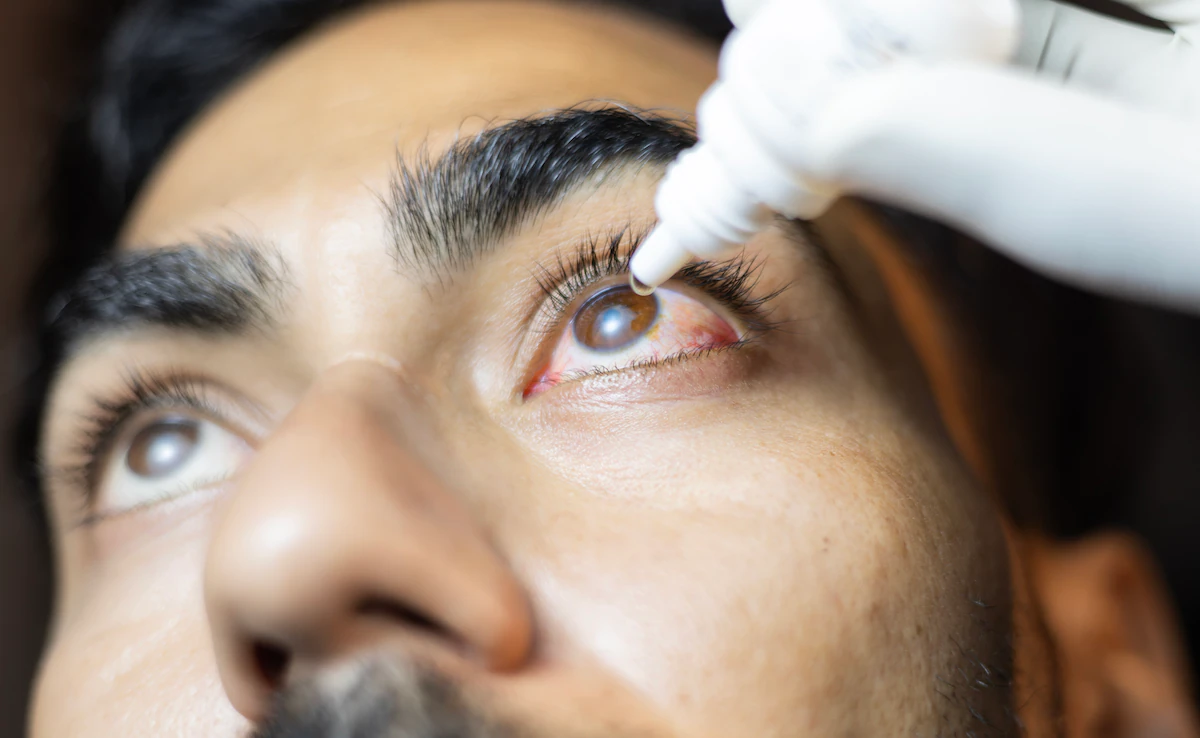
New Delhi: India’s drug regulator has suspended Entod Pharmaceutical Ltd.’s authorization to manufacture and market a specific eye drop product, citing the company’s unapproved promotional claims. The Drugs Controller General of India (DCGI) issued the suspension order on September 10, addressing concerns that the eye drops were marketed as reducing the need for reading glasses…
-
Restoring memories: Breakthroughs in treating dementia

Exciting Advances in Dementia Research Revealed at 4th Asia Summit on Global Health The 4th Asia Summit on Global Health (ASGH) on May 16th showcased promising developments in dementia research, offering hope for patients and their families. Dementia, characterized by the loss of memory, speech, and identity, is a growing global concern. However, recent medical…